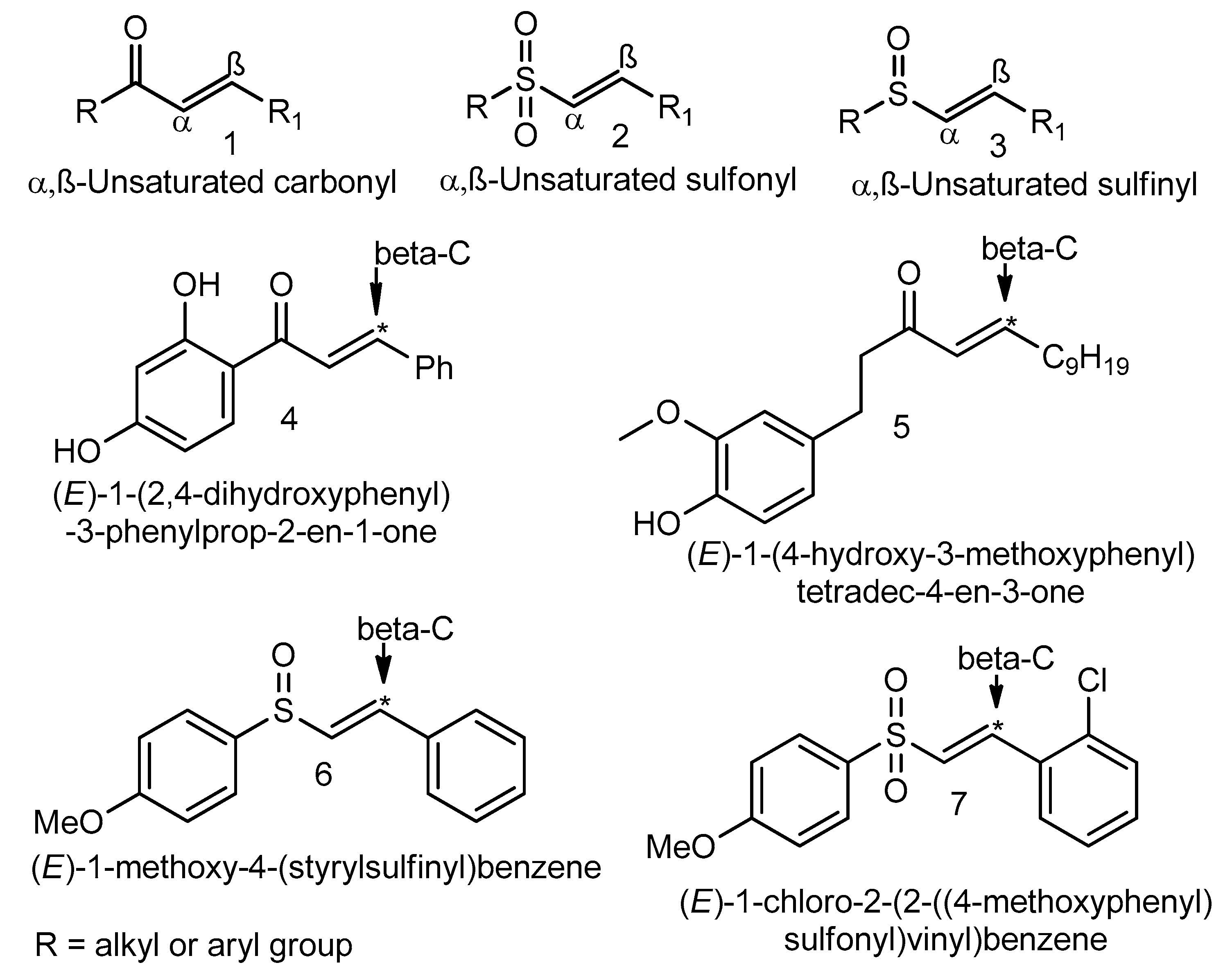
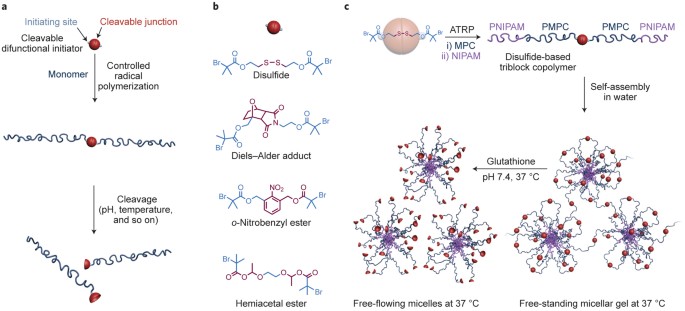
Microwave-Assisted Palladium-Catalyzed Cross-Coupling of Aryl and Vinyl Halides with H-Phosphonate Diesters
SOLVED: Draw all resonance structures for vinyl acetate? Determine which hydrogens have more electron density on the alkene moiety?

Recent advances in applications of vinyl ether monomers for precise synthesis of custom-tailored polymers - ScienceDirect
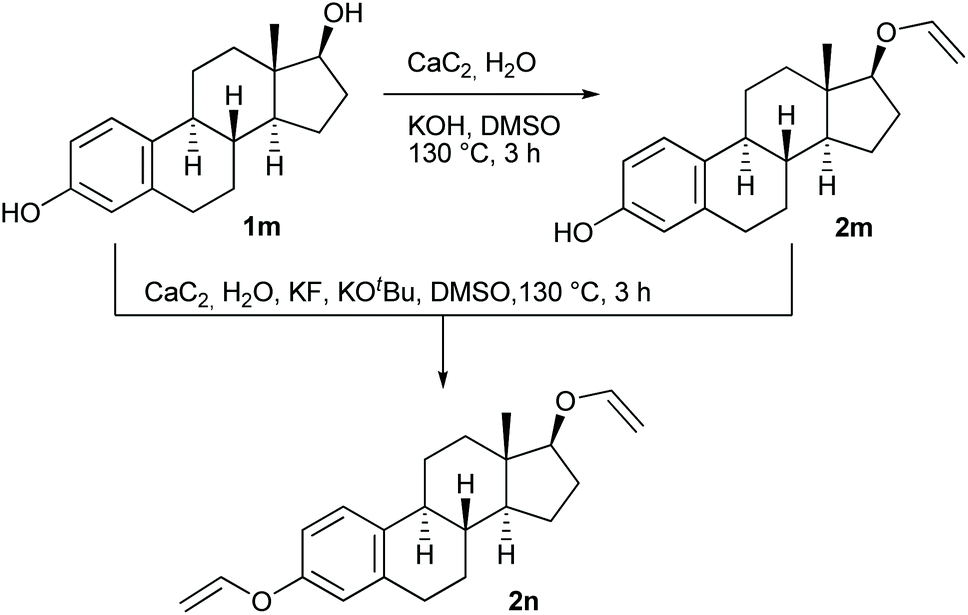
Examining the vinyl moiety as a protecting group for hydroxyl (–OH) functionality under basic conditions - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D0QO00202J
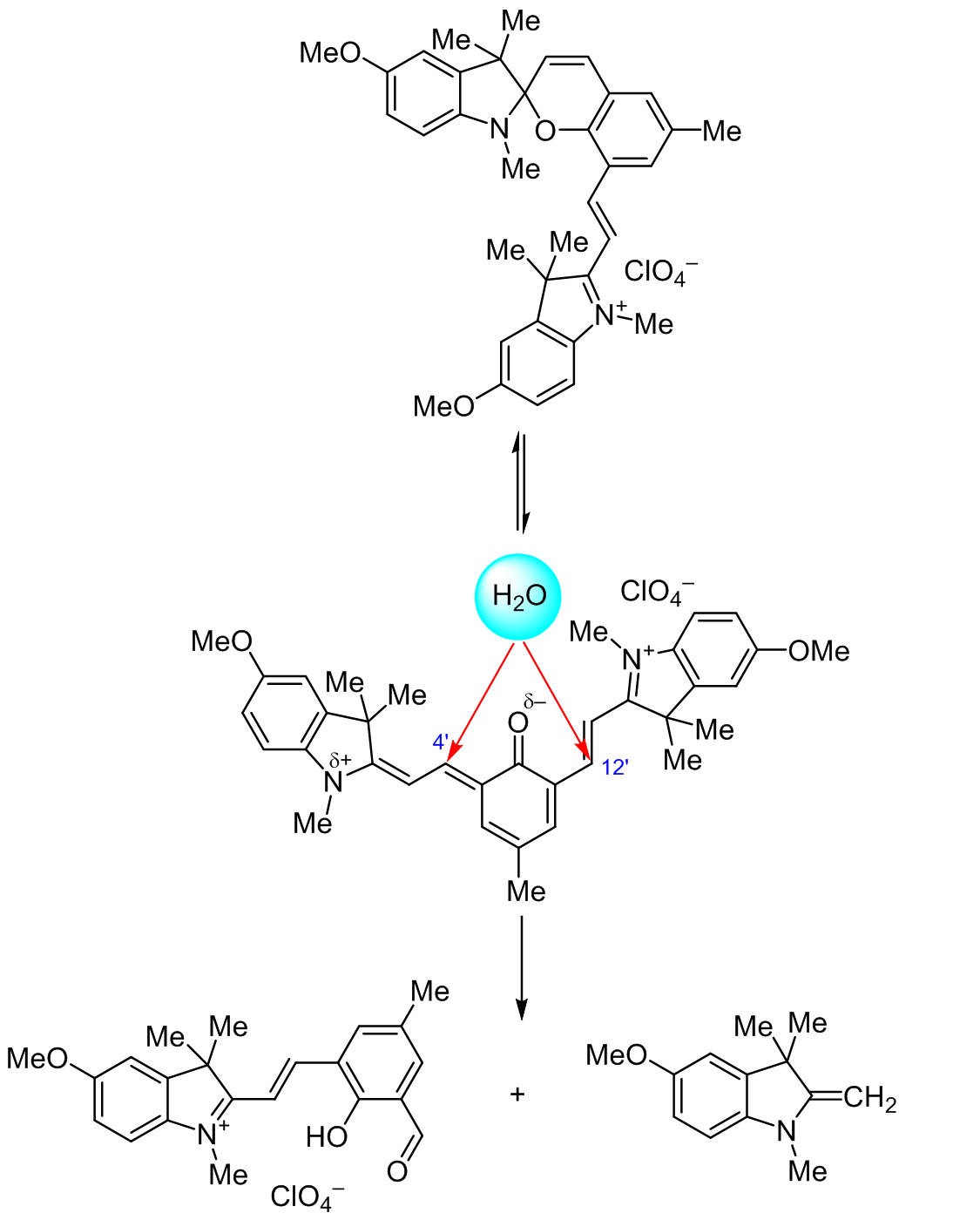
SYNTHESIS AND STRUCTURAL CHARACTERIZATION OF NEW SPIROPYRAN CONTAINING CONJUGATED VINYL-3<i>Н</i>-INDOLIUM MOIETY AND ITS HYDROLYSIS PRODUCT | Козленко | Chemistry of Heterocyclic Compounds
![Poly[di(ethylene glycol) vinyl ether]-stabilized poly(vinyl acetate) nanoparticles with various morphologies via RAFT aqueous emulsion polymerization of vinyl acetate | Polymer Journal Poly[di(ethylene glycol) vinyl ether]-stabilized poly(vinyl acetate) nanoparticles with various morphologies via RAFT aqueous emulsion polymerization of vinyl acetate | Polymer Journal](https://media.springernature.com/full/springer-static/image/art%3A10.1038%2Fs41428-020-00417-3/MediaObjects/41428_2020_417_Figa_HTML.png)
Poly[di(ethylene glycol) vinyl ether]-stabilized poly(vinyl acetate) nanoparticles with various morphologies via RAFT aqueous emulsion polymerization of vinyl acetate | Polymer Journal

The Vinyl Moiety as a Handle for Regiocontrol in the Preparation of Unsymmetrical 2,3‐Aliphatic‐Substituted Indoles and Pyrroles - Huestis - 2011 - Angewandte Chemie - Wiley Online Library

Structure-activity relationships of cephalosporins having a (dimethylisoxazolidinio)vinyl moiety at their 3-position. | Semantic Scholar
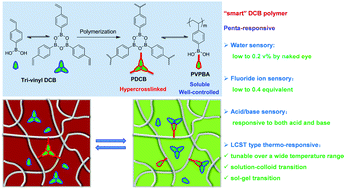
Well-controlled polymerization of tri-vinyl dynamic covalent boroxine monomer: one dynamic covalent boroxine moiety toward a tunable penta-responsive polymer - Polymer Chemistry (RSC Publishing)

Radical polymerization behavior and thermal properties of vinyl ethylene carbonate derivatives bearing aromatic moieties - ScienceDirect
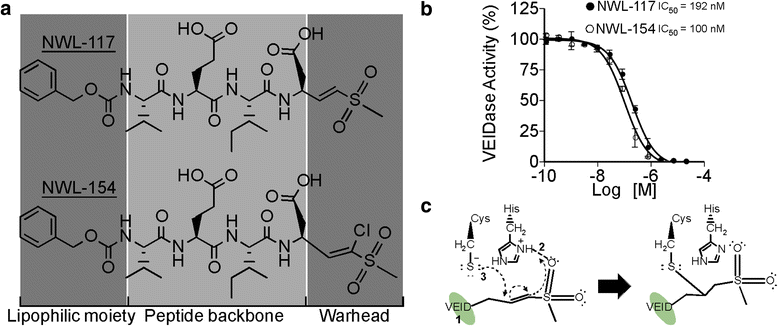
Caspase vinyl sulfone small molecule inhibitors prevent axonal degeneration in human neurons and reverse cognitive impairment in Caspase-6-overexpressing mice | Molecular Neurodegeneration | Full Text
![PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f286ceb376de35cacaa09ad45f90c27e91883fe4/3-Table1-1.png)
PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar

A Versatile New Monomer Family: Functionalized 4-Vinyl-1,2,3-Triazoles via Click Chemistry | Journal of the American Chemical Society

Examining the vinyl moiety as a protecting group for hydroxyl (–OH) functionality under basic conditions - Organic Chemistry Frontiers (RSC Publishing)

Vinyl Cation Stabilization by Silicon Enables a Formal Metal‐Free α‐Arylation of Alkyl Ketones - Pons - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Polymers | Free Full-Text | Tin Complexes Containing an Atenolol Moiety as Photostabilizers for Poly(Vinyl Chloride)

Controlled Radical Homopolymerization of Representative Cationically Polymerizable Vinyl Ethers | Journal of the American Chemical Society