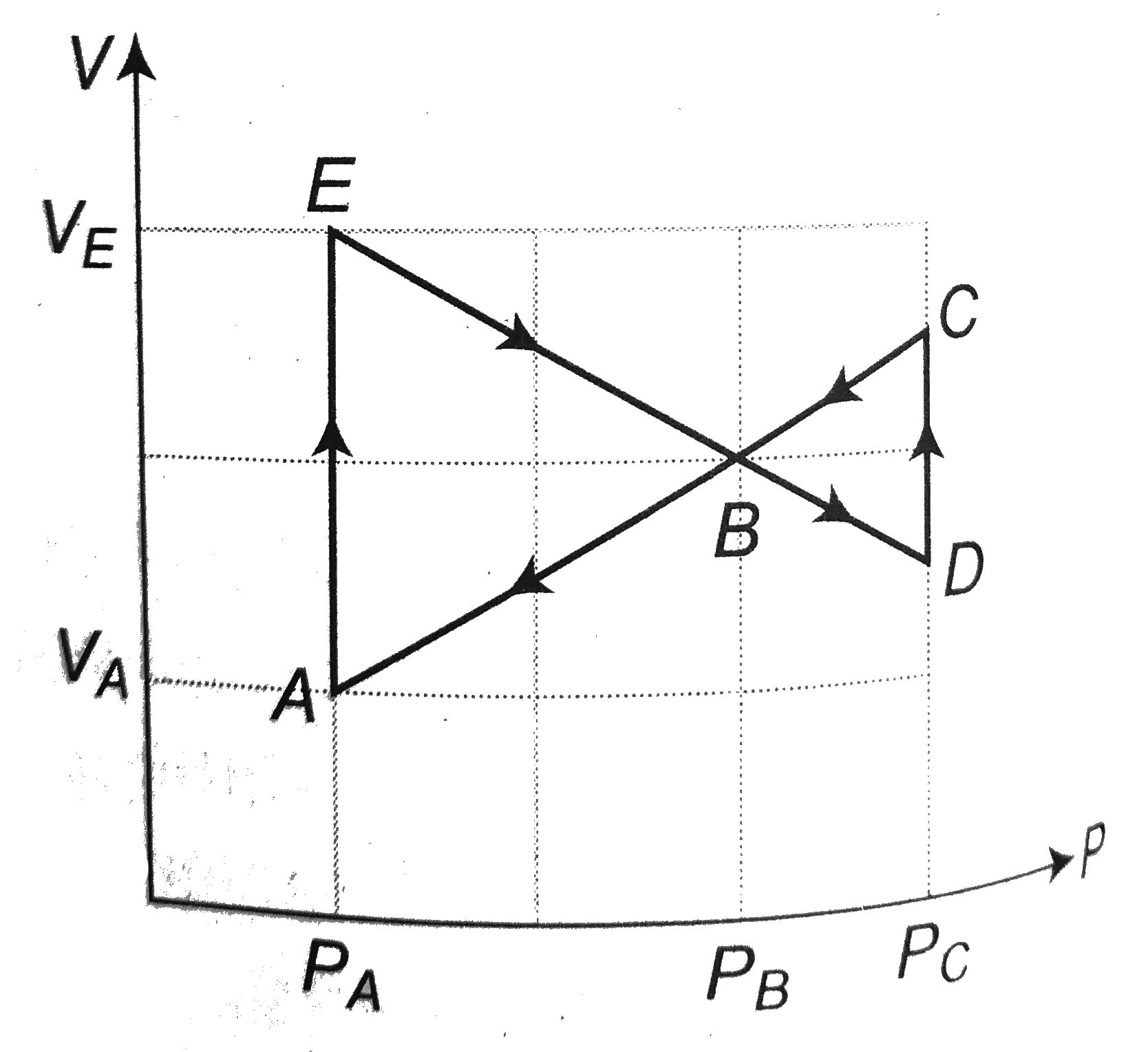
SOLVED: Consider 3 mol of monoatomic ideal gas that is taken Irom stale A (Pa 2 atm. 10 L) to state B (Pr = atm: VR 30 L)as shown below Pa =

Gases Pressure (5.1) Gas Laws of Boyle, Charles, Avogadro (5.2) Ideal Gas Law (5.3) Gas Stoichiometry (5.4) Kinetic Molecular Theory (5.6) Effusion & Diffusion. - ppt download







:max_bytes(150000):strip_icc()/sun-beams-diffuse-ocean-micronesia-palau-128941208-587b91ad5f9b584db36312c9.jpg)