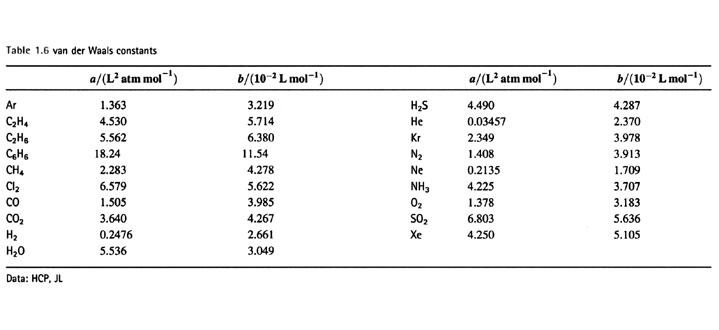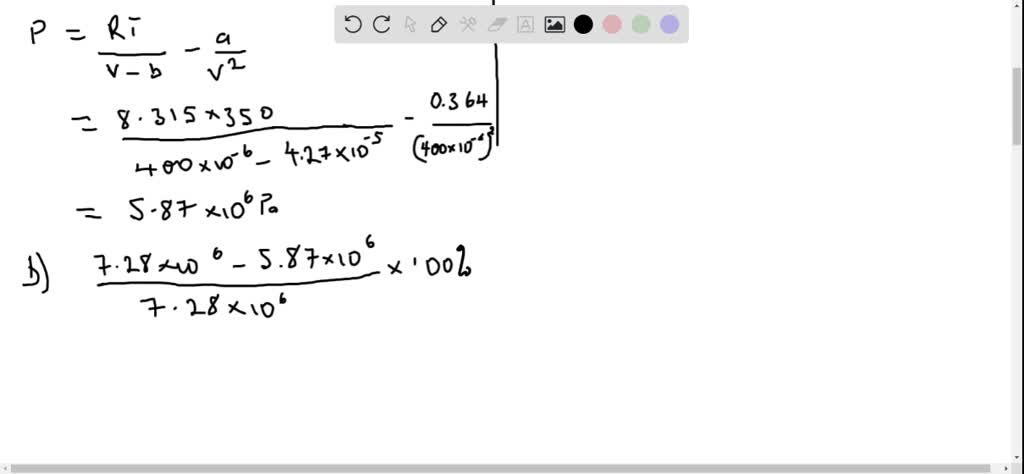
SOLVED:Deviations from the Ideal-Gas Equation. For carbon dioxide gas (CO2), the constants in the van der Waals equation are a=0.364 J ·m^3 / mol^2 and b=4.27 ×10^-5 m^3 / mol . (a)

Solving Van der Waals Equation of State in Microsoft Excel using Newton Raphson Method (Part 2) - YouTube
![SOLVED: (a) [5 marks] Plot a graph of compression factor (y-axis) vs pressure (X-axis) for an ideal gas. Oz, CH4, and CzH4 as real gases obeying the van der Waals equation of SOLVED: (a) [5 marks] Plot a graph of compression factor (y-axis) vs pressure (X-axis) for an ideal gas. Oz, CH4, and CzH4 as real gases obeying the van der Waals equation of](https://cdn.numerade.com/ask_images/11fd4350a67a4a928ae6428255b08864.jpg)
SOLVED: (a) [5 marks] Plot a graph of compression factor (y-axis) vs pressure (X-axis) for an ideal gas. Oz, CH4, and CzH4 as real gases obeying the van der Waals equation of

Lec 20 | ChemE Thermo | Demonstration of PV relationship plotted using VdW equation of state - YouTube

I need an analytical expression to solve for the change in pressure with temperature at constant volume for ammonia at

Next-Generation Nonlocal van der Waals Density Functional | Journal of Chemical Theory and Computation





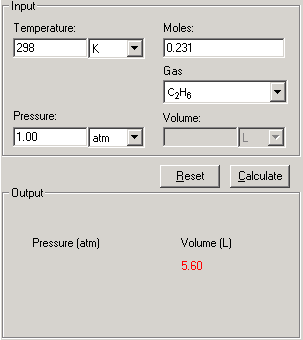
![Solved] Calculate the pressure that CCl4 will exe | SolutionInn Solved] Calculate the pressure that CCl4 will exe | SolutionInn](https://s3.amazonaws.com/si.question.images/image/images11/876-(568).png)