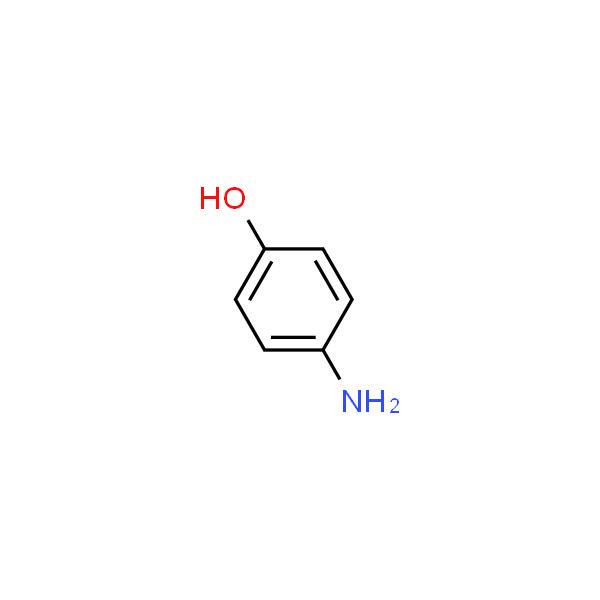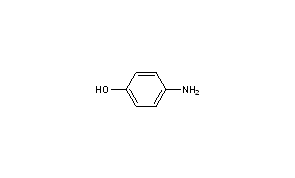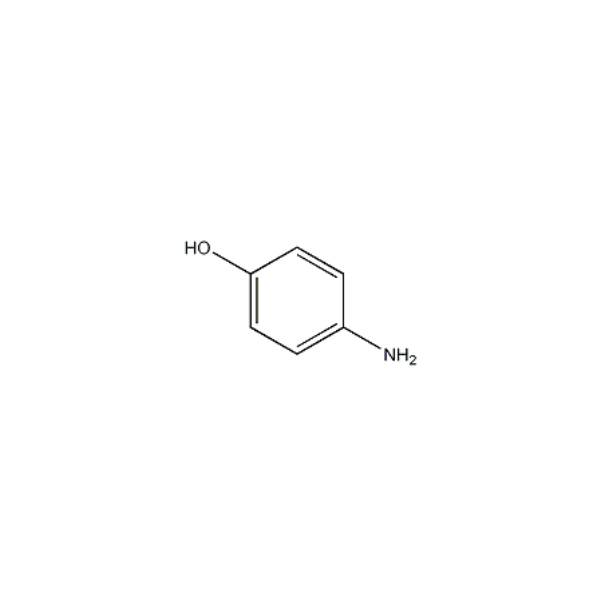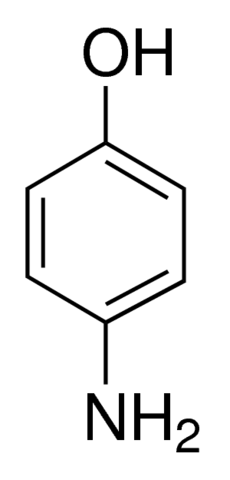
CAS-123-30-8, p-Aminophenol Pract (4-Aminophenol) Manufacturers, Suppliers & Exporters in India | 025911
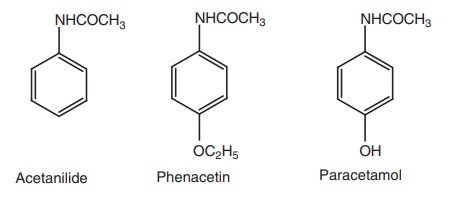
p-Amino phenol derivatives - SAR, Structure, Properties, uses, Synthesis, Assay, Storage, Dosage forms, Dose | Synthesis and Drug Profile

Synthesis of p-Aminophenol by Catalytic Hydrogenation of p-Nitrophenol | Organic Process Research & Development

Figure 13. Oxidation of p-Aminophenol : Organic Redox-Flow Batteries Using Hair Dyes and Pharmaceuticals : Science and Education Publishing

Genotoxic permanent hair dye precursors p-aminophenol and p-toluenediamine electrochemical oxidation mechanisms and evaluation in biological fluids - ScienceDirect

In the reaction p-aminophenol + acetic anhydride arrow acetaminophen + acetic acid, is it true that acetic acid is formed as a by-product by the reaction between the unused acetyl group of
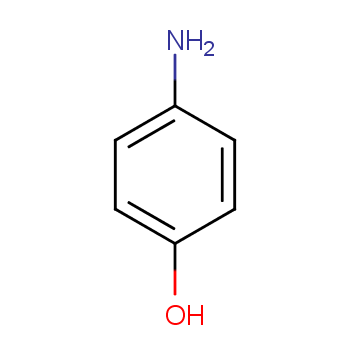
High quality 4-Aminophenol/p-Aminophenol Cas 123-30-8 with specialized manufacturer 123-30-8 Purity 99% 25kg/drum or as you required China

p - aminophenol reacts with one equivalent of acetyl chloride in the presence of pyridine to give mainly.

Which of the following statements are true regarding the reaction between p- aminophenol and acetic anhydride. 1. As the reaction proceeds, p-aminophenol loses a hydrogen ion from its amine group. 2. As the