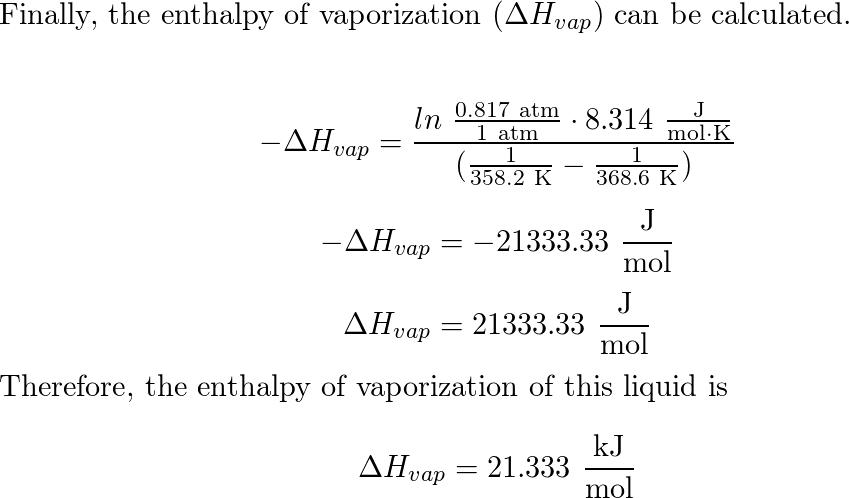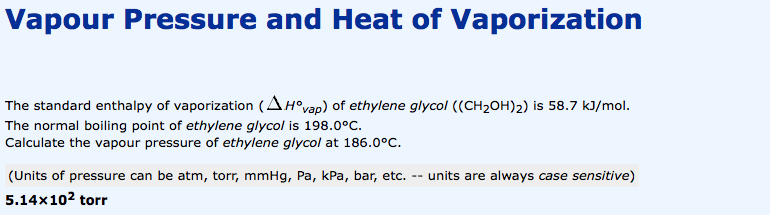E N T H A L P Y - H heat content in matter: E N T H A L P Y - H heat content in matter: natural systems tend to go from a state of high energy to a. - ppt download
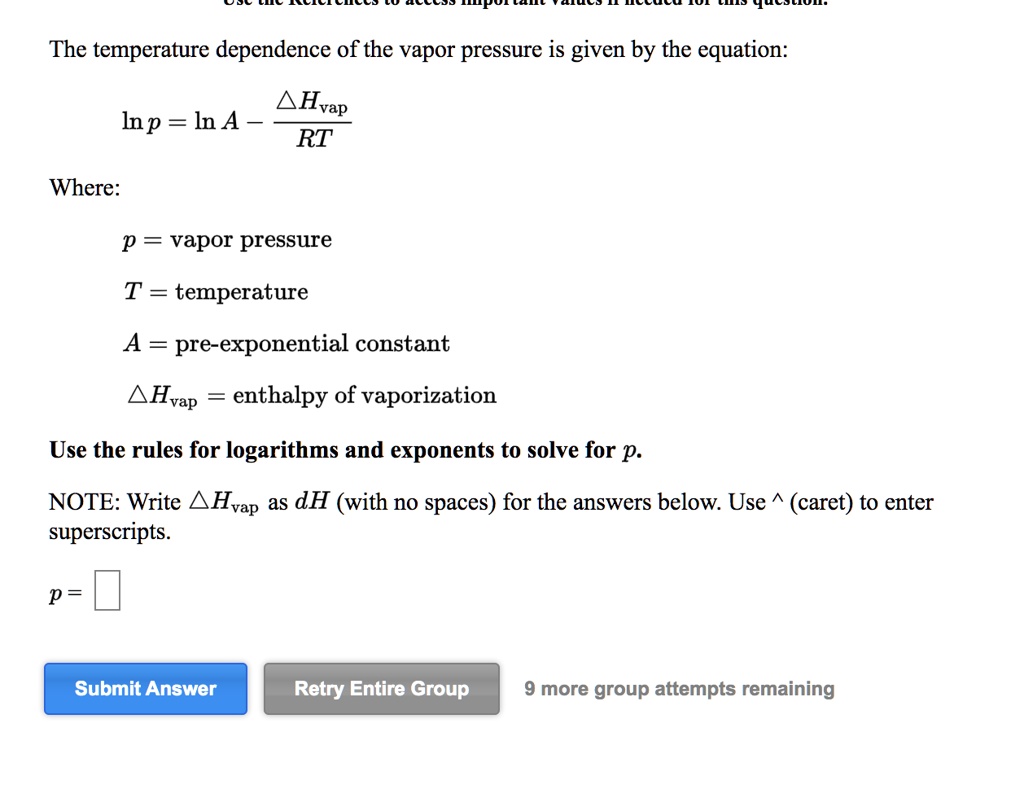
SOLVED: The temperature dependence of the vapor pressure is given by the equation: Hvap Inp InA - RT Where: p = vapor pressure T = temperature pre-exponential constant Hvap enthalpy of vaporization

Heat of vaporization ∆H vap as a function of temperature at pressure... | Download Scientific Diagram

SOLVED:What is the ΔH vap ^∘ of a liquid that has a vapor pressure of 621 torr at 85.2^∘ C and a boiling point of 95.6^∘ C at 1 atm ?

∆H vap predictions of polymer RE by MLR and KRR (d = 1), ARE for MLR:... | Download Scientific Diagram

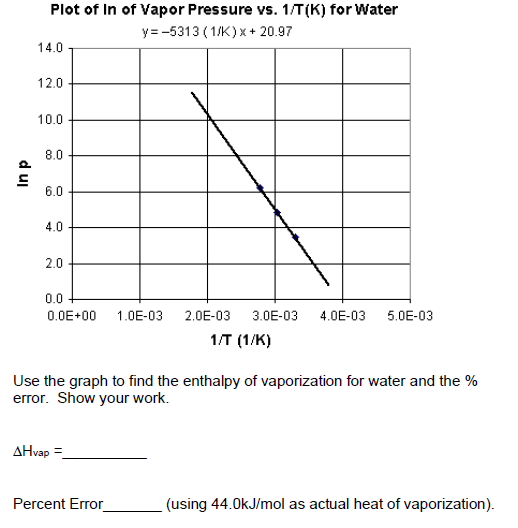
thermodynamics - $\Delta \bar{h}_{vap}$ and $\Delta \bar{s}_{vap}$ from vapor pressure vs. temperature data - Chemistry Stack Exchange
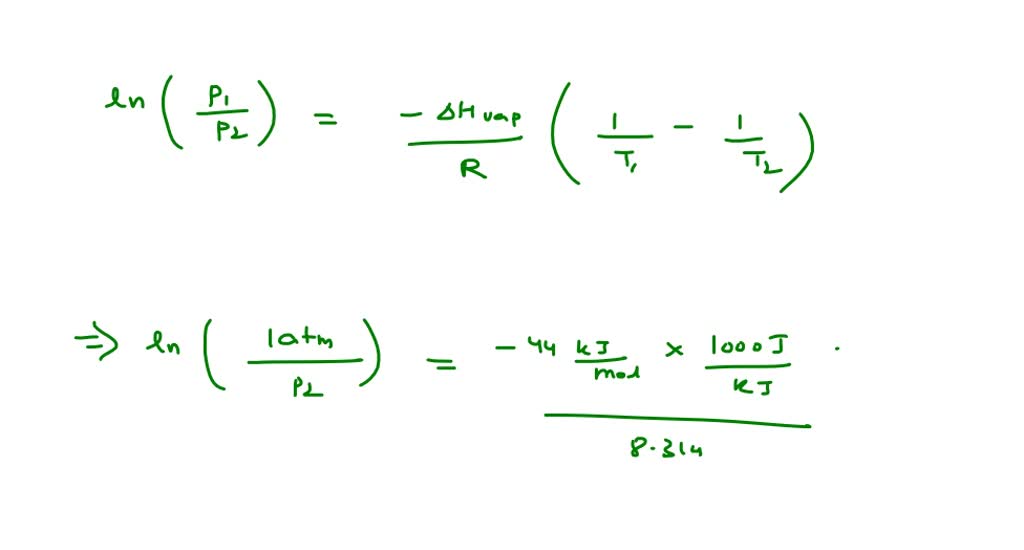
SOLVED: The standard enthalpy of vaporization (H°vap) of ethylenediamine ((NH2CH2)2) is 44.0 kJ/mol. The normal boiling point of ethylenediamine is 116.5°C. Calculate the vapour pressure of ethylenediamine at 95.0°C.

Clausius – Clapeyron Equation This equation is a relation between H vap and pressure at a certain Temperature. - ppt download
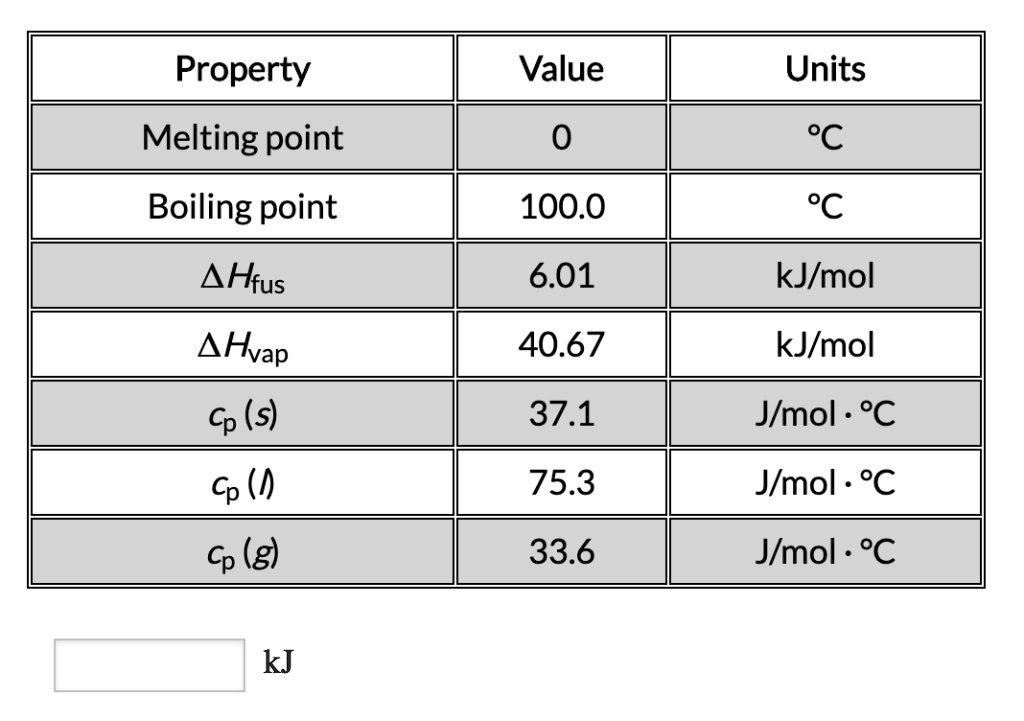
SOLVED: Property Melting point Boiling point Hfus Hvap (s) Value Units 0 100.0 6.01 kJlmol 40.67 kJlmol 37.1 Jlmol (h 75.3 Jmol . *C Cp (g) 33.6 Jlmol - kJ









-438.png)