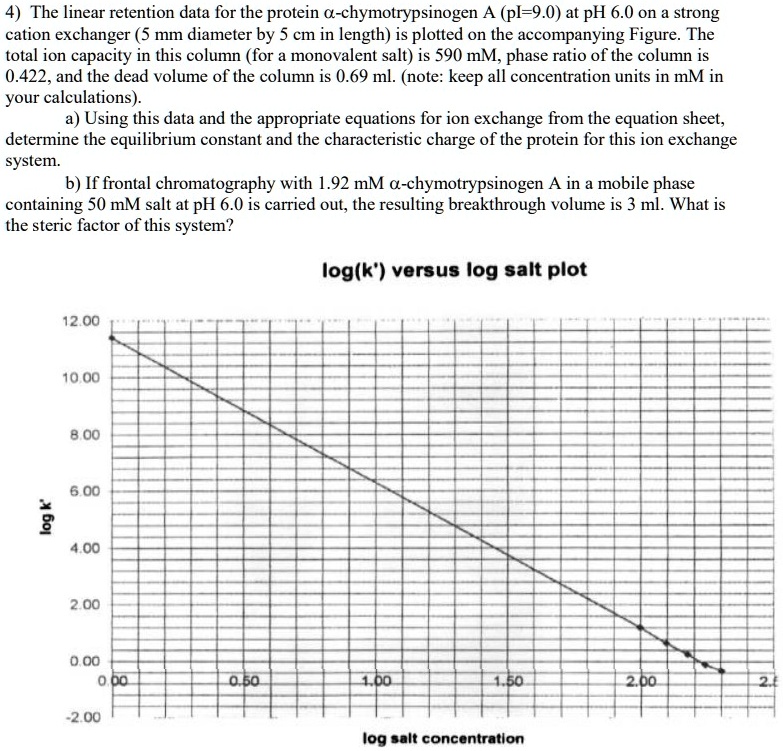
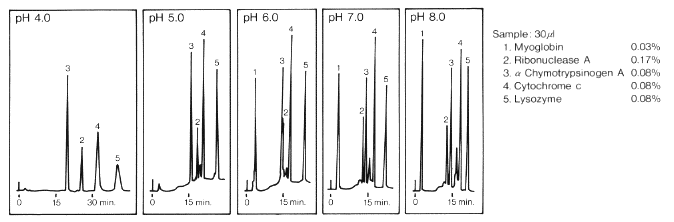
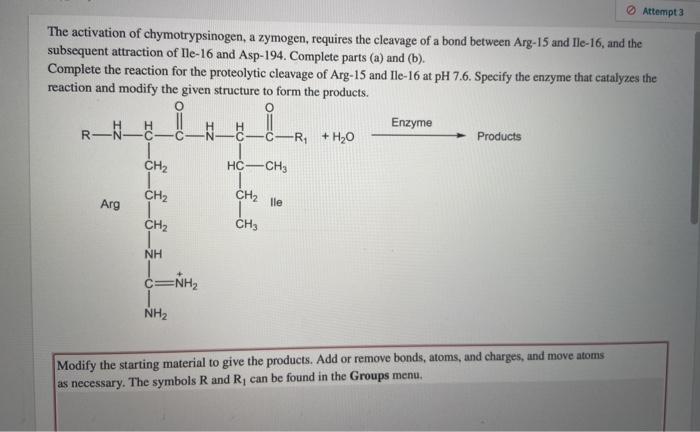
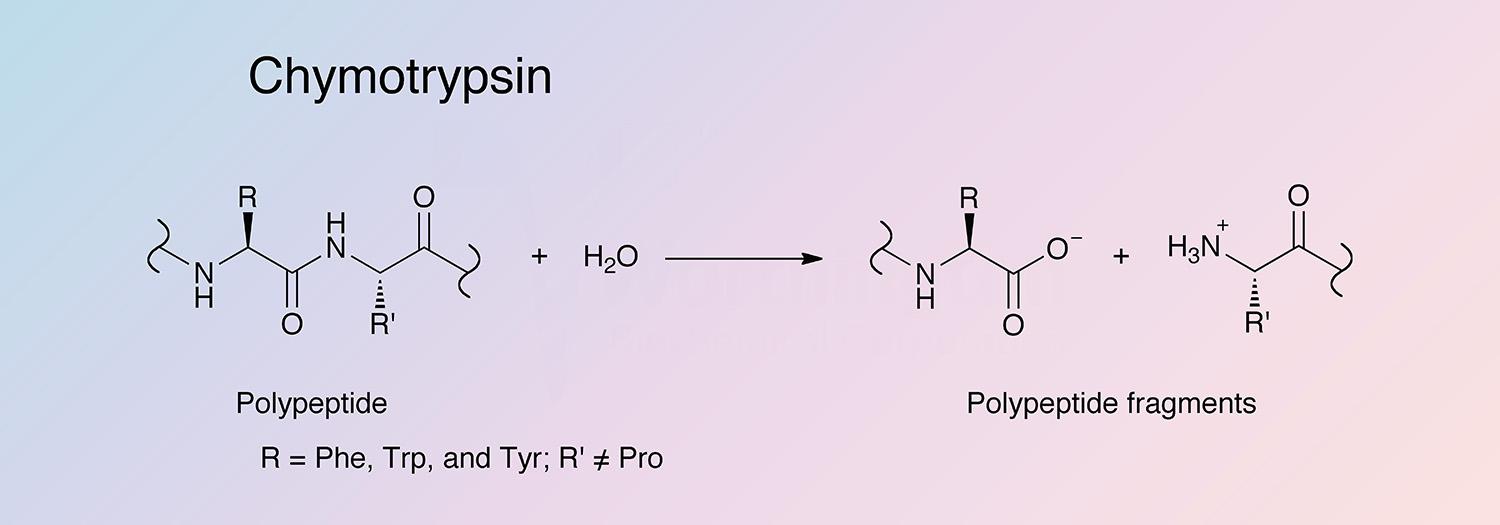
Relating particle formation to salt‐ and pH‐dependent phase separation of non‐native aggregates of alpha‐chymotrypsinogen a - Journal of Pharmaceutical Sciences

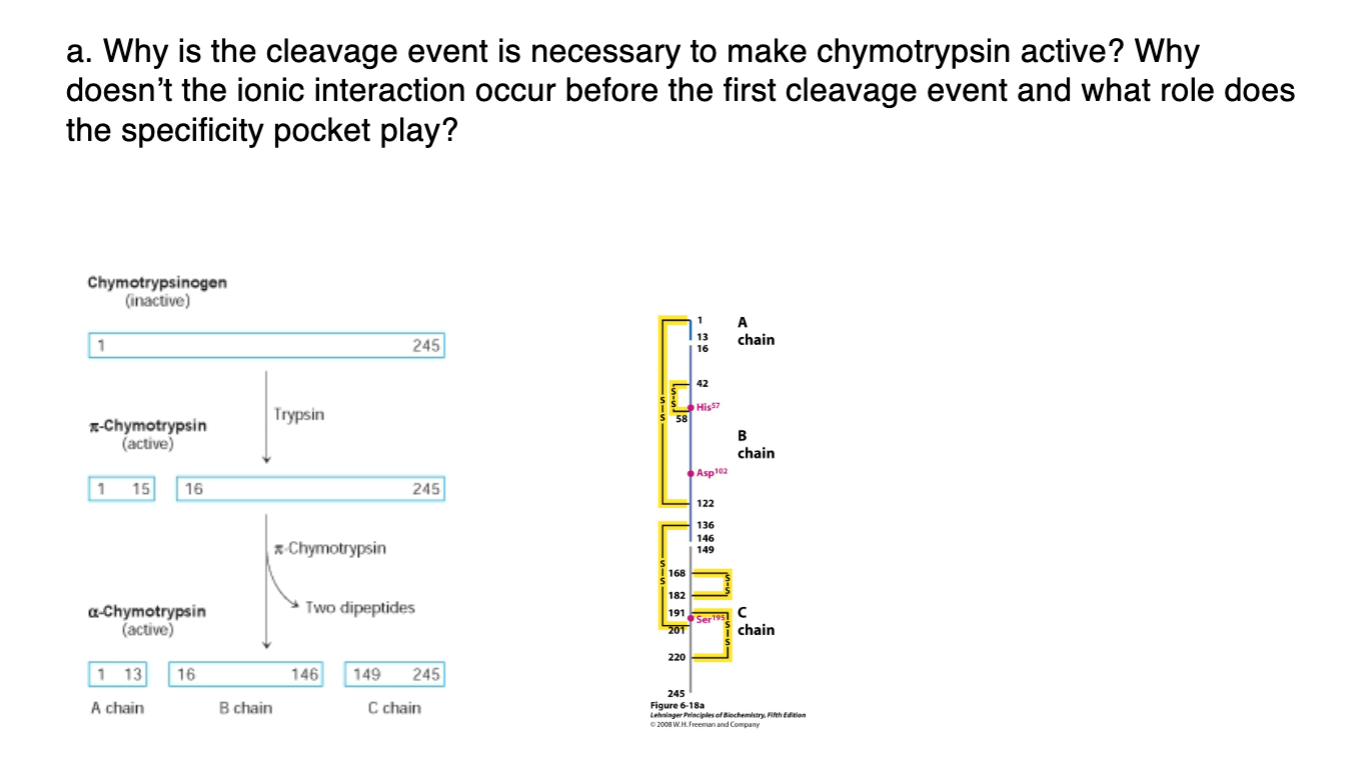
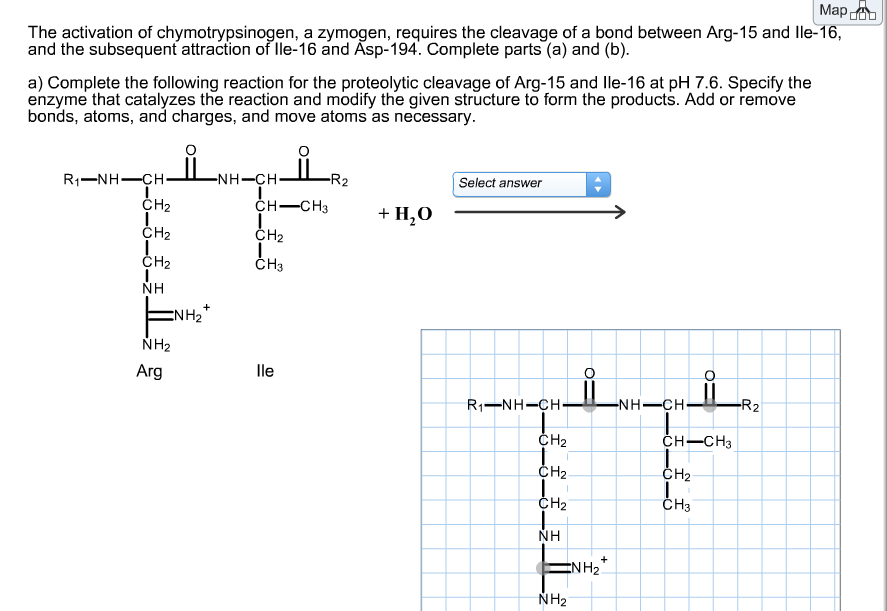
Studies of the chymotrypsinogen family. IV. The conversion of chymotrypsinogen A to alpha-chymotrypsin. | PNAS

SOLVED: The isoelectric point, pI, of the protein chymotrypsinogen A is 9.1 while that of peroxidase is 7.2 What is the net charge of chymotrypsinogen at pH 5.1 What is the net

Engineered ChymotrypsiN for Mass Spectrometry-Based Detection of Protein Glycosylation | ACS Chemical Biology

Aggregation of α-Chymotrypsinogen A in Aqueous Solutions Aaron Aziz Advisor: Dr. Yun Liu Colleague: Dr. Jiang Du. - ppt download
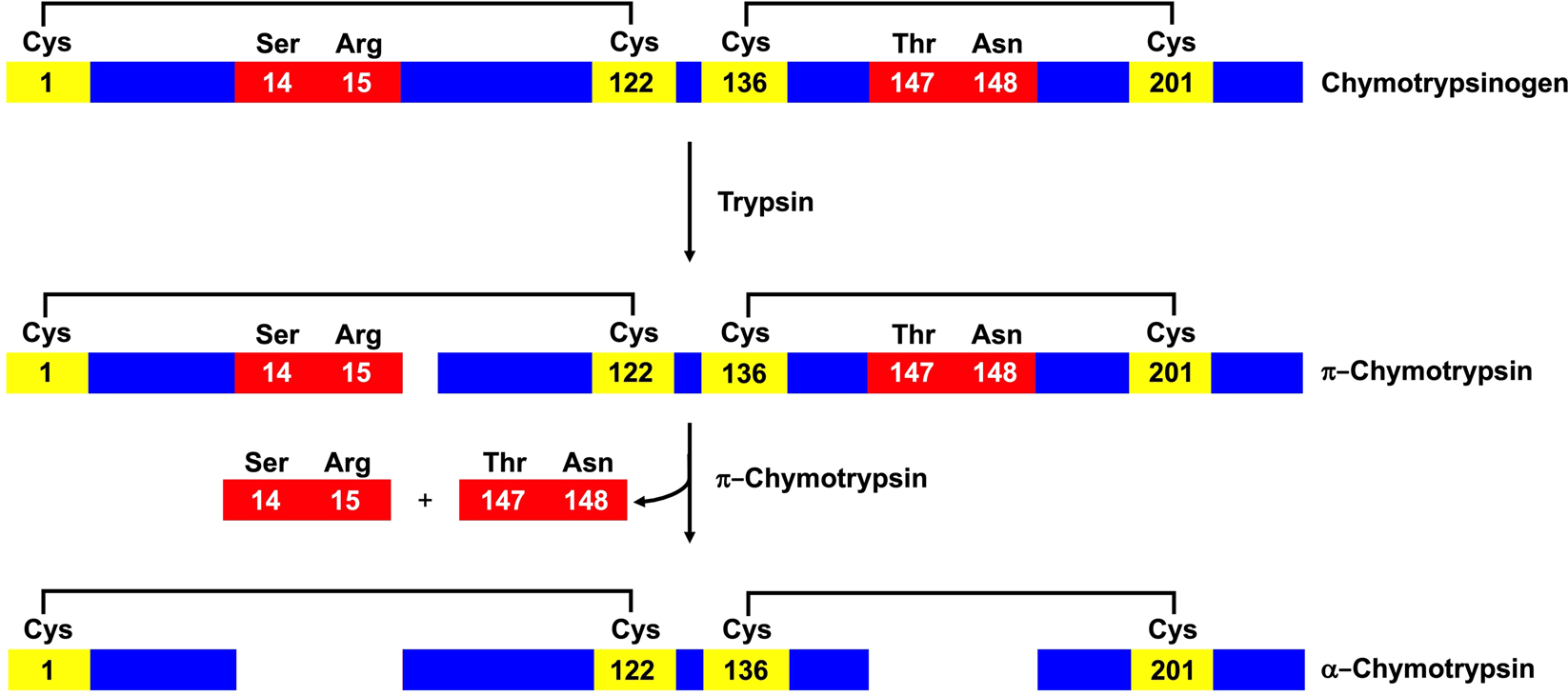
Ultra-rapid glutathionylation of chymotrypsinogen in its molten globule-like conformation: A comparison to archaeal proteins | Scientific Reports

RCSB PDB - 2CGA: BOVINE CHYMOTRYPSINOGEN A. X-RAY CRYSTAL STRUCTURE ANALYSIS AND REFINEMENT OF A NEW CRYSTAL FORM AT 1.8 ANGSTROMS RESOLUTION
1CHG: Chymotrypsinogen,2.5 Angstroms Crystal Structure, Comparison With Alpha-Chymotrypsin,And Implications For Zymogen Activation

Macromolecular crowding stabilises native structure of α-chymotrypsinogen-A against hexafluoropropanol-induced aggregates - ScienceDirect