Using the data presented in the pressure-versus-volume drawing below (x = 1.00E+4 Pa and y = 2.00E-3 m^3), calculate the magnitude of the work done when the system changes from A to
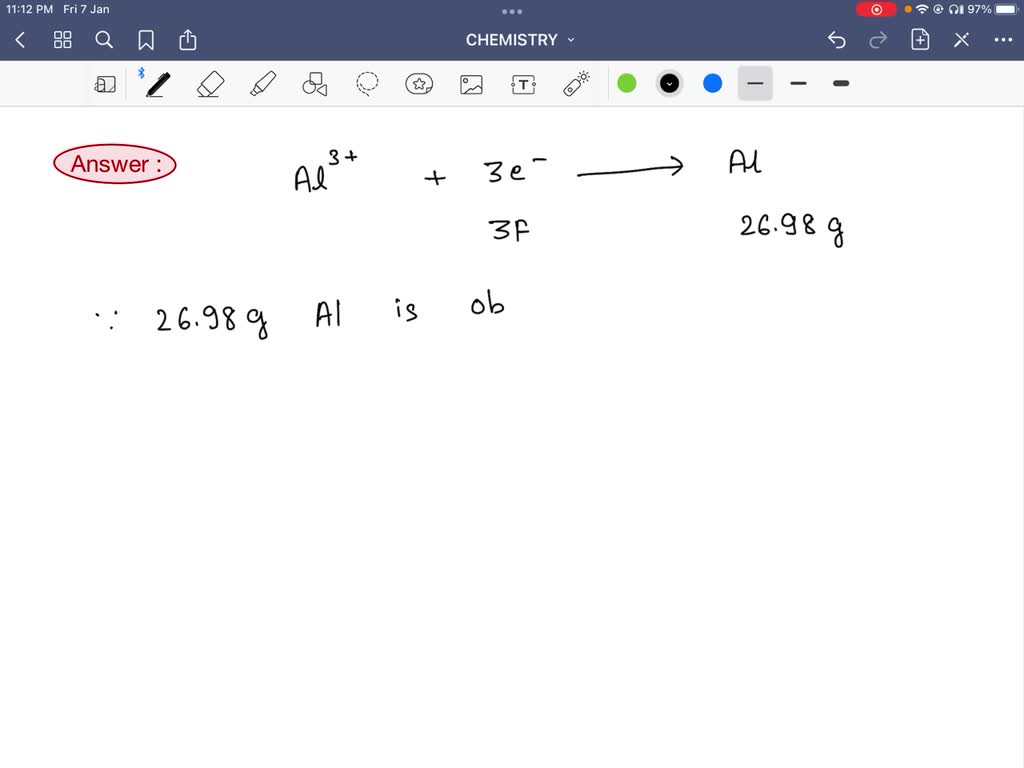
SOLVED: 13. How many faradays are required to reduce 1.00 g of aluminum(III) to the aluminum metal? (a) 1.00 (b) 1.50 (c) 3.00 (d) 0.111 (e) 0.250 Please show solution how to derive answer. Thank you
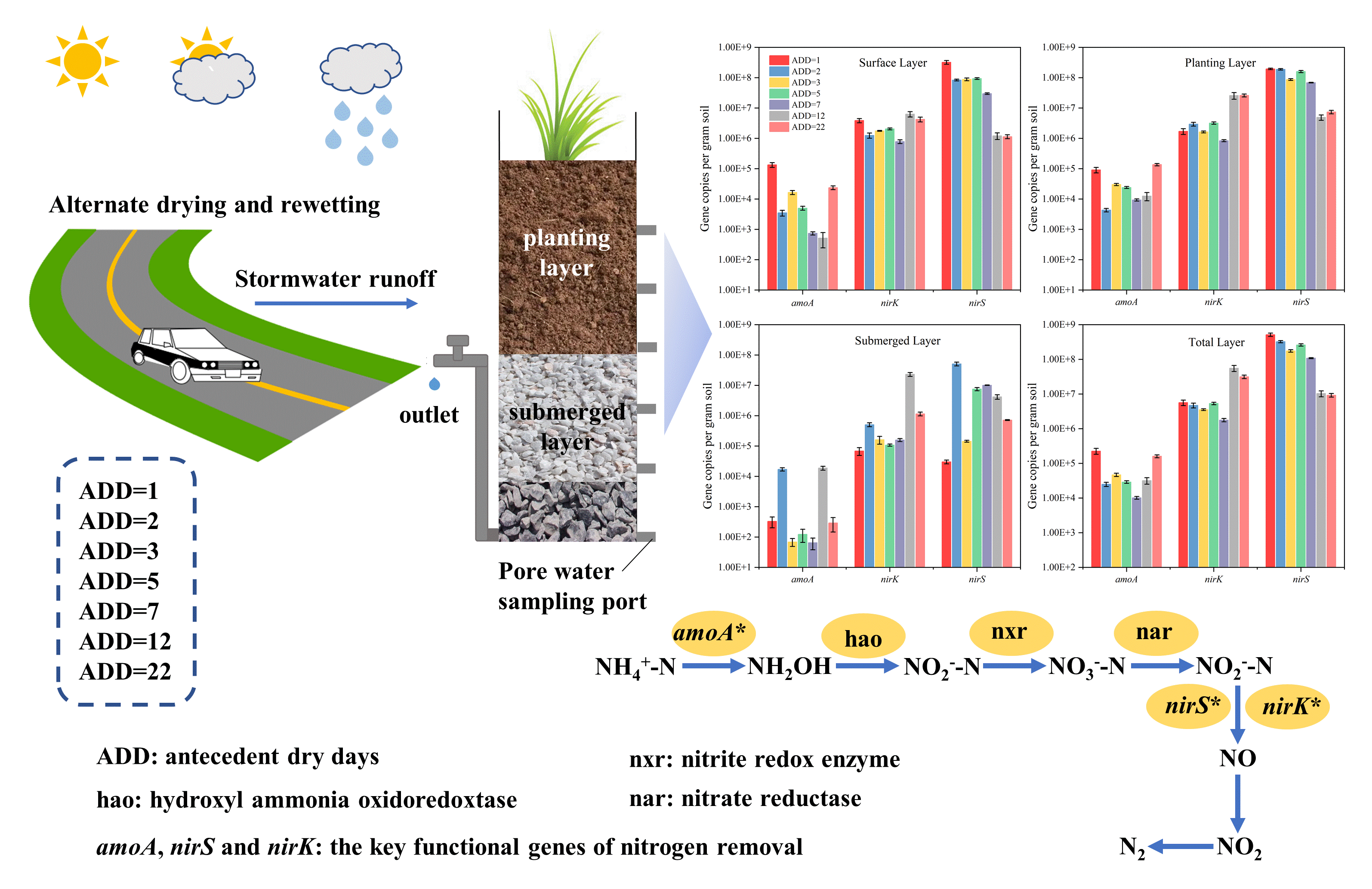
Nitrogen process in stormwater bioretention: Effect of the antecedent dry days on the relative abundance of nitrogen functional genes
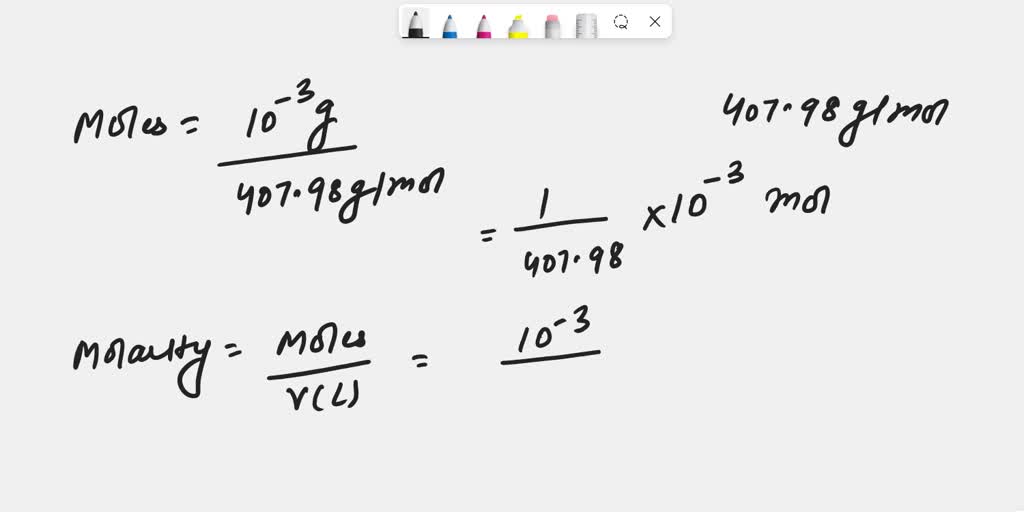
SOLVED: 1. Calculate the Molarity of 1.00E-3% mass crystal violet solution assuming at this concentration the density of the solution is 1.00 g/mL and the molar mass of crystal violet is 407.98
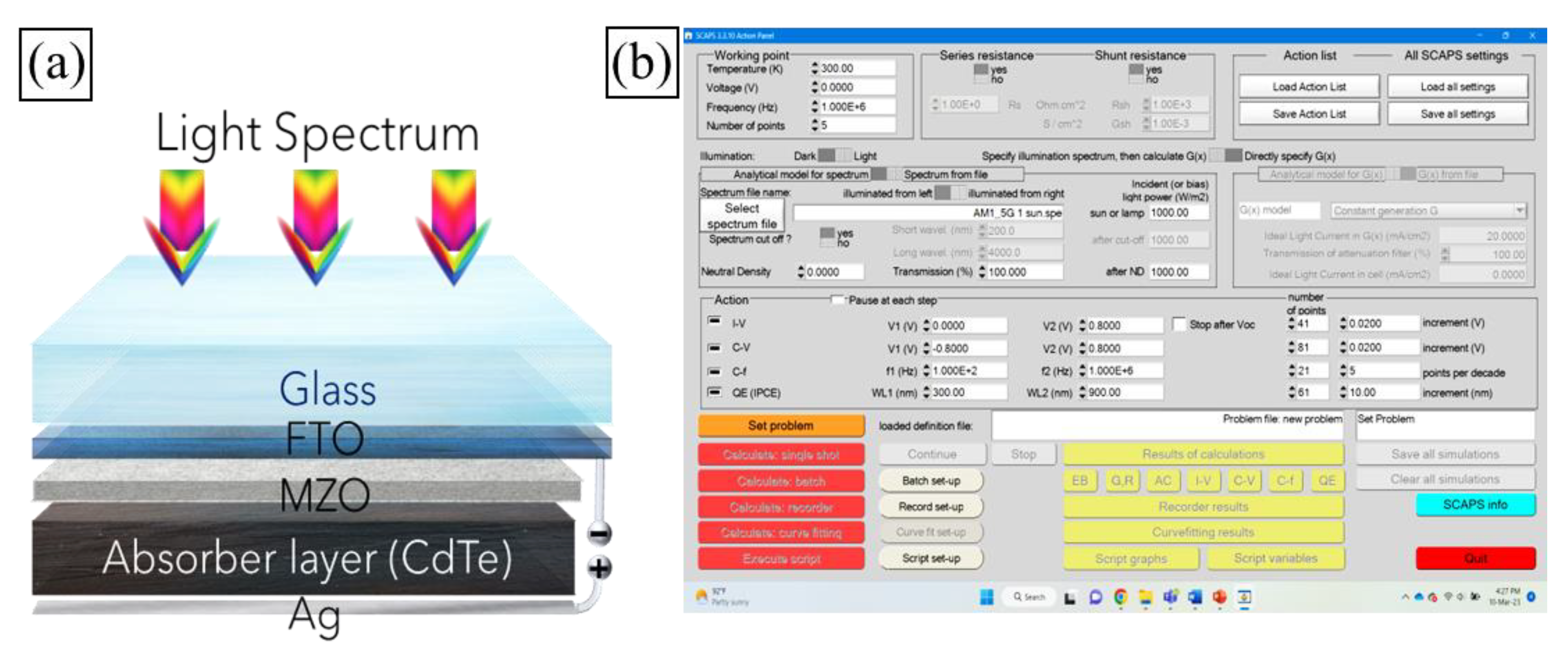
Crystals | Free Full-Text | Effect of Cu2Te Back Surface Interfacial Layer on Cadmium Telluride Thin Film Solar Cell Performance from Numerical Analysis








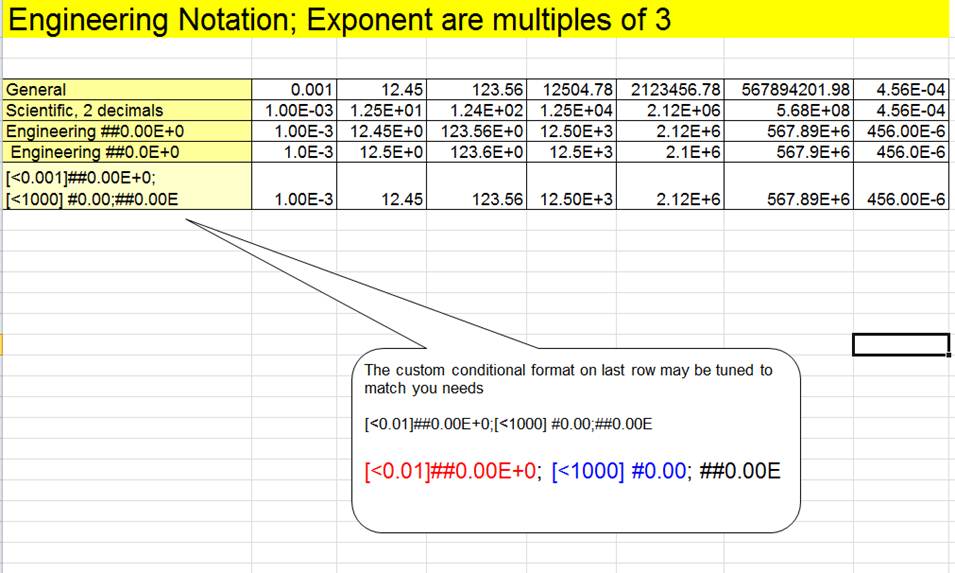
![Excel2019 10の2乗や3乗等のべき乗表示をする方法について[指数表示] | find366 Excel2019 10の2乗や3乗等のべき乗表示をする方法について[指数表示] | find366](https://find366.com/wp-content/uploads/2021/01/2021-01-16_21h53_52-640x309.png)
![駿河屋 -<中古>1.00-E-0008[R]:[煌めくステージへ]丸山彩(アニメ・ゲーム) 駿河屋 -<中古>1.00-E-0008[R]:[煌めくステージへ]丸山彩(アニメ・ゲーム)](https://www.suruga-ya.jp/database/pics_light/game/gg829326.jpg)